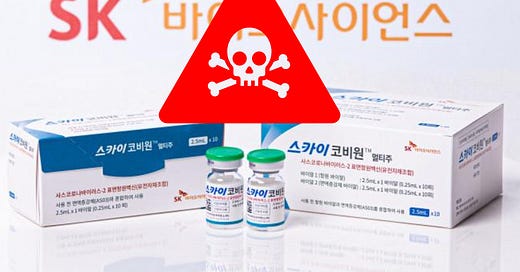
This is quite an alarming developing story that not enough people are paying attention to. With thanks to Debi Evans for this information.
In the segment starting at 47 mins on UK Column News (31 May 2023) Debi Evans discusses the new SKYCovion covid vaccine that has just got regulatory approval from the MHRA “after meeting the MHRA’s required safety, qu…
Keep reading with a 7-day free trial
Subscribe to Where are the numbers? by Norman Fenton and Martin Neil to keep reading this post and get 7 days of free access to the full post archives.