The smoking man emails
Was there ever a deadly and novel virus?
Those of you familiar with the cult 90s TV series the X-files will recall the role of the smoking man, who like ‘deep throat’ in the Watergate scandal, would reveal snippets of the truth to Mulder and Scully at critical points in their shared adventures.
Back in 2020 I had my very own smoking man. He was anonymous but I called him “The Cleric”. We started conversing by email around September 2020, after I published some articles in Toby Young’s Lockdown Sceptics (LDS) website.
He sent me some long emails about the origins story of the so-called pandemic with a special focus on the virus and PCR testing. Much of it I couldn’t understand at the time, and some of it I will admit I still struggle with even today because, as you all know neither Norman nor I are “wetware” scientists - we do software and statistics not human biology or virology or pharmacology or whatever. Given this we try to be careful not to make fools of ourselves and stay in our lanes.
For all I knew The Cleric might have sent these emails off to many of the movers and shakers in our ‘movement’. I have no idea. But I didn’t really know what to do with his information. Obviously, I shared it in the small circle of sceptics who were coalescing around seeking answers to open questions about the virus and PCR testing at the time: Mike Yeadon, Clare Craig, Scott McLachlan and a few others. However, I didn’t get much traction, and with hindsight this was probably because we were all grappling together in the dark to some extent, looking for the same key, but each searching in different directions. Plus given I’m a biological dunce I probably wasn’t too persuasive.
I have posted The Cleric’s revelations below, and in summary they covered:
The role of Dr Christain Drosten and the Sars-cov2 virus genomic sequencing
The dodgy PCR testing that resulted and the cycle problem (which you will all be familiar with and probably be a little bored by)
The Instand report, with acknowledgment to Drosten, which confirmed that PCR test being used in labs worldwide were generating false positives of circa 9%.
Revealing cross reactivity and non-specificity of the PCR test, so that it picked up other coronaviruses (and the human genome!?)
A 9% PCR false positive rate sounds pretty high, doesn’t it? Much higher than even sceptics had been postulating at the time (which was typically no higher than 5%). To put that in Bayesian context this would mean that if, say 1 in 1000 people were genuinely infected with the Sars-cov2 virus, then a person testing positive will almost certainly (98.9% probability) NOT have the virus. And with zero Sars-cov2 virus prevalence nobody testing positive has the virus, but we’d still see 9% of those tested, testing positive. So, logically, you wouldn’t necessarily need a deadly novel virus to give the world the impression of the widespread presence of said virus. But something had to be causing the false positives, and if it wasn’t a deadly novel coronavirus then what was it? Well, the cross reactivity and non-specificity of the PCR test maybe tells us the answer: other coronaviruses and perhaps other pathogens.
At that point in September 2020 no credible voice was shouting to the world that “there is no novel & deadly coronavirus” and I certainly couldn’t persuade anyone of this, being a humble numbers guy. And to be totally honest I wasn’t sure I believed it either. So, I parked The Cleric’s information at the back of my mind and moved on to other things (the UK government were producing a self-replenishing wall of shit data to deal with, which warranted my attention). And anyway, as I said, I could not get anyone properly qualified really engaged.
The next chapter in the story developed in early 2021 when I discovered that the UK government had been conducting bogus PCR testing in late 2020 and used it to push the covid wave in the run up the launch of the injections. Peter Doshi helped me get this scandal in the BMJ as a rapid response letter. Basically, I revealed that the false positive rate for covid PCR testing was much higher than anyone could ever imagine in their worst nightmares - up to 65% of positive tests were false positives, by the ONS’s own published statistics. However, this wasn’t caused by high PCR cycles but was because the laboratories abandoned using the WHO guidelines and manufacturer standards for testing. What did they do? Rather than require two out of three gene positives to define a positive for covid, they decided only one was enough!
Details here:
This evidence again pointed to cross reactivity and non-specificity of the PCR test. It looked to be designed to pick up, well, anything that you might want it to detect. So, rather than find residue of Sars-cov2 virus fragments perhaps it seemed to be quite brilliant at finding any coronavirus. Hence, maybe the test worked perfectly well but just not for the purpose, or in the way they told us it worked.
Personally, I thought this a bit of a bombshell. But when I published the BMJ letter I didn’t get much traction and when I alerted the world on twitter only gained a few hundred impressions. LDS were good enough to publish it, for which I am grateful. And, sure, colleagues on our side thought it a scandal but I got the impression that for them it looked like it was a piece of the jigsaw that didn’t fit the emerging picture before us at the time. Collective focus remained on the lab leak versus Wuhan market theories and our attention then switched to the vaccines, for understandable reasons.
Now many of us are looking back at Spring 2020 and are revisiting some of the origins of the covid-19 debacle. There is renewed focus on iatrogenic harms resulting from policy and fresh and pertinent questions about whether the virus was ever novel and deadly. This might seem like ‘raking over old coals’ but finding answers to these questions is as important as ever. Because, if our understanding of what happened is wrong the covid nightmare will surely repeat itself in future.
So, without much further ado, and without any comment from me, here are the emails from ‘The Cleric’. I urge you all to read it, share and discuss in the comments.
Note: subsequent to first publication of this article a helpful commentor has pointed out to me that the Instand report mentioned here is available in English from here.
From The Cleric
The reason they can get away with this is because, despite claims that the debate around the pandemic reaction were driven by cold, hard numbers and statistics, it really isn't, instead it's showing a glaring lack of evidence in numbers, which is the reason why Mr Hancock can claim in a live segment that the PCR test results are accurate up to 99.9% (or was it 99.99%?) and the other side won't challenge the assertion because they don't have hard evidence at hand.
It's time to change that.
1. Dr Drosten, infamous inventor of abusing RT-PCR tests for "clinical diagnosis" of an "infection" with SARS-CoV-2, set a cycling threshold (CT) of 45 for his blueprint screening system for SARS-CoV-2:
https://www.eurosurveillance.org/content/10.2807/1560-7917.ES.2020.25.3.2000045#html_fulltext
Thermal cycling was performed at 55 °C for 10 min for reverse transcription, followed by 95 °C for 3 min and then 45 cycles of 95 °C for 15 s, 58 °C for 30 s.
As James Ferguson has shown here:
https://lockdownsceptics.org/radical-uncertainty-and-government-innumeracy/
a CT of 45 is way beyond the threshold for detecting viable virus in samples:
It has been known since April, that even apparently heavy viral load cases “with Ct above 33-34 using our RT-PCR system are not contagious and can thus be discharged from hospital care or strict confinement for non-hospitalized patients.” A review of 25 related papers by Carl Heneghan at the Centre for Evidence-Based Medicine (CEBM) also has concluded that any positive result above 30Ct is essentially non-viable even in lab cultures (i.e. in the absence of any functional immune system), let alone in humans. However, in the US, an amplification of 40Ct is common (1 trillion copies) and in the UK, COVID-19 RT-PCR tests are amplified by up to 42Ct. This is 2 to the power of 42 (i.e. 4.4 trillion copies), which is 4,400x the ‘safe’ screening limit. The higher the amplification, the more likely you are to get a ‘positive’ but the more likely it is that this positive will be false.
Dr Drosten's work is the foundation for every RT-PCR screening kit for SARS-CoV-2 on the market. The more sensible labs in Germany are lowering this to a 30-32 CT, but Drosten is openly inviting labs in his paper to go as high as 45, and apparently US labs are welcoming this, as the New York Times has recently shown.
2. Dr Drosten recently claimed that the false positive rate is insignificant because lab work is so exceptional that incorrect results don't pass, the reason being that all labs performing RT-PCR tests must be certified. Yes, the labs might be certified - but not the test kits. In fact, no RT-PCR screening kit on the market ever received any kind of approval or certification (not to be confused with emergency authorization in the US, which I'll come back to in a bit). The EMA, which happens to be headquartered in Sweden, never approved a single RT-PCR screening kit because those are not medical products and therefore do not require approval before distribution.
So what do they require?
https://ec.europa.eu/info/sites/info/files/testing_kits_communication.pdf
I won't quote the document in full, but basically manufacturers self-certify the kit with a CE marking, pretty much the same way a sweatshop in any corner of the world would manufacture christmas tree lighting for a few pennies and slap a CE marker on it with a single-page declaration of conformity, and call it a day.
3. There is an officially-designated organisation in Germany that's regularly organising a voluntary specificity and sensitivity auditing of RT-PCR screening kits for SARS-CoV-2. The most recent report from June 2020 is here:
I'm sure you'll be able to find a colleague or assistant from Germany who can translate this for you.1
This is not rogue work by lockdown sceptics, but officially sanctioned by the federal government, and officially supported by the Charite Berlin, Dr Drosten's employer and the designated institution in Germany for coordinating SARS-CoV-2 research. 453 labs from 36 countries were participating in the study. Basically every lab in the study was sent blind positive and negative samples, and they were asked to perform tests with a SARS-CoV-2 screening kit of their choice and report back results, e.g. "we believe sample A is positive, sample B is negative, ...". Results were collated into the kits used. The paper doesn't list the name of the lab, but it does list which screening kit was used. Unless labs were bored and willing to spend money for nothing, it's safe to assume that participants are actually part of each country's official testing effort.
Have a look at pages 12 to 13. Column C denotes the expected test result for the supplied sample, and column E denotes how many samples were identified correctly. Column F shows the average cycle thresholds used.
Pages 24 and following break it down to screening kits used, and detailed results for primers.
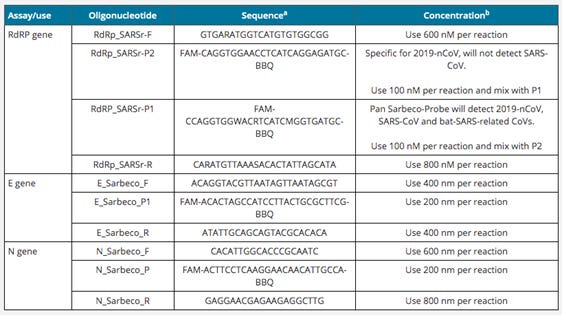
In case you are not familiar with the RT-PCR process, and jumping back to Drosten's paper linked above for a moment, have a look at this table:
The RdRP assays identify sequences in the SARS-CoV-2 RNA (or so Dr Drosten claims even though he never isolated the virus himself and never had access to the isolated virus but instead downloaded the virus RNA sequence from a database, as he admits in the paper, and which he attributes to Chinese researchers who later said that the isolated virus sample became unusable shortly after uploading the sequence so they destroyed all remains), while the E gene and N gene assays are controls against relatives of SARS-CoV-2 in the larger family of bats.
Pages 24 and following in the study list results for E gene, N gene, RdRP gene and ORF1a(b) primers. Including results for E gene, N gene primers, and ORF1a(b) primers (for SARS-CoV-1 RNA), i.e. control primers that detect relatives of SARS-CoV-2, is skewing the statistics, of course.
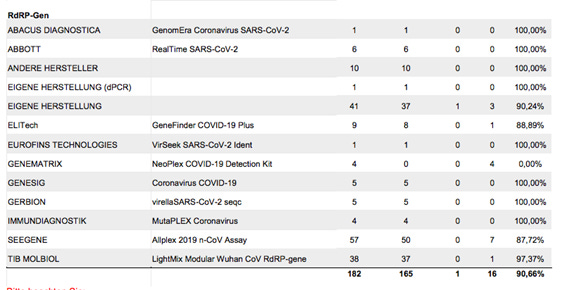
Have a look at page 50:
These are results for the negative sample, using the RdRP primers specific to SARS-CoV-2. 182 negative samples were tested, 165 were correctly identified as negative, 1 as indeterminate, and 16 of the negative samples were incorrectly identified as positive. Resulting in a false positive rate of 9.34% for this primer set.
Of course one needs to look at the aggregate result in the hope that labs would mark the complete throat swab sample as negative when just one set of primers tests positive, but the others test negative. In the current climate where "scientists" such as Dr Whitfield and Dr Vallance manufacture facts at will, that's a big ask. Actually, there are reports in German media (mainstream, not hobbyist blogs) that certified labs, of which Dr Drosten is so fond of, have been marking throat swabs as positive even in cases where they ran out of some reagents (for detecting primer sequences) and therefore couldn't perform the whole test suite, and where just a few primers were detected.
4. The FDA, with litigation laws in the US being a bit more dangerous to vendors than in the EU, is approaching this more carefully. Here is the list of all RT-PCR screening kits that have received emergency use authorization (EUA) so far:
This is not FDA approval. The FDA is not vouching for anything.
The latest entry is for Thermo Fisher's TaqPath COVID-19 Combo Kit. When you click on the title it'll show you the text of the EUA:
https://www.fda.gov/media/136113/download
Quotes, emphasis mine:
Your product is authorized for the qualitative detection of nucleic acid from SARS-CoV-2 in upper respiratory specimens (such as nasopharyngeal, oropharyngeal, nasal, and mid-turbinate swabs, and nasopharyngeal aspirate) and bronchoalveolar lavage (BAL) specimens from individuals suspected of COVID-19 by their healthcare provider. (...)
I have concluded, pursuant to Section 564(d)(2) of the Act, that it is reasonable to believe that the known and potential benefits of your product, when used for the qualitative detection of SARS-CoV-2 and used consistent with the Scope of Authorization of this letter (Section II), outweigh the known and potential risks of your product. I have concluded, pursuant to Section 564(d)(3) of the Act, based on the totality of scientific evidence available to FDA, that it is reasonable to believe that your product may be effective in diagnosing COVID-19, when used consistent with the Scope of Authorization of this letter (Section II), pursuant to Section 564(c)(2)(A) of the Act. (...)
No descriptive printed matter, including advertising or promotional materials, relating to the use of your product may represent or suggest that this test is safe or effective for the detection of SARS-CoV-2.
Notice the contradiction regarding effectiveness. All EUAs on the page feature similar wording. There is a "funny" mistake in the EUA for Thermo Fisher where it reads, emphasis mine:
No descriptive printed matter, including advertising and promotional materials, relating to the use of your product shall clearly and conspicuously state that:
* This test has not been FDA cleared or approved;
It should read something like this:
https://www.fda.gov/media/135661/download
All advertising and promotional descriptive printed matter relating to the use of the authorized New York SARS-CoV-2 Real-time RT-PCR Diagnostic Panel shall clearly and conspicuously state that:
* This test has not been FDA cleared or approved;
Which makes sense. As the "emergency" in EUA clearly indicates, this was an emergency action to have something, really anything, to track the pathogen's spread. Instead, politicians across the globe are misconstruing the tests as hard proof of an infection, when the FDA clearly states that it's not.
So how effective is this particular test by Thermo Fisher?
Thermo Fisher Scientific’s conclusion from investigations of customer complaints indicate that inadequate vortexing or centrifugation can lead to false positive results. Thermo Fisher Scientific has updated these instructions to reduce the risk of inaccurate results. The updated instructions related to vortexing and centrifugation are important for both laboratories performing testing according to the authorized instructions for use and laboratories who are performing validated modifications outside of the authorization.
The kit received EUA on March 13th, this note by the FDA was released on August 17th. Surely the CDC has recalled and adjusted official numbers to account for all false positives generated by Thermo Fisher's kit in the past 5 months.
There are more notes like the above, for example:
Emphasis mine:
The U.S. Food and Drug Administration (FDA) is alerting clinical laboratory staff and health care providers of an increased risk of a false positive result with BD SARS-CoV-2 Reagents for the BD Max System test. In one study, the manufacturer found approximately three percent (3%) of results were false positive results.
Not quite the 0.1% that Mr Hancock was claiming.
5. The FDA has also been busy measuring specificity and sensitivity of antibody tests on the market:
Notice this paragraph, emphasis mine:
Tests are also described by their Positive and Negative Predictive values (PPV and NPV). These measures are calculated using a test's sensitivity, its specificity, and using an assumption about the percentage of individuals in the population who have antibodies to SARS-CoV-2 (which is called "prevalence" in these calculations). Every test returns some false positive and false negative results. The PPV and NPV help those who are interpreting these tests understand, given how prevalent individuals with antibodies are in a population, how likely it is that a person who receives a positive result from a test truly does have antibodies to SARS-CoV-2 and how likely it is that a person who receives a negative result from a test truly does not have antibodies to SARS-CoV-2. The PPV and NPV of a test depend heavily on the prevalence of what that test is intended to detect. Because all tests will return some false positive and some false negative results, including tests that detect antibodies to SARS-CoV-2, broad use of the tests, when not appropriately informed by other relevant information, such as clinical history or diagnostic test results, could identify too many false-positive individuals.
With the UK and Germany set on gradually extending the RT-PCR testing regime with antibody tests "to track the spread of the virus", no doubt the next few months will see an exponential growth of "cases".
Addendum:
As I mentioned previously, Dr Drosten, lead author of the initial paper sparking the RT-PCR frenzy for detecting SARS-CoV-2, did not have access to the isolated virus when he devised his method. As the paper points out:
All oligonucleotides were synthesised and provided by Tib-Molbiol (Berlin, Germany).
Which means that the reagents of the RT-PCR screening kit for SARS-CoV-2 were targeting RNA sequences (i.e. oligonucleotides) expressly synthesised, i.e. manufactured by Tib-Molbiol. (NB: The paper was amended in July to state that there was a conflict of interest because one of the authors of the paper happens to be the CEO of Tib-Molbiol, Germany's largest and most "successful" vendor of RT-PCR screening kits for SARS-CoV-2.) In other words: the screening kit can correctly identify synthetic RNA sequences made for this kit.
In the absence of isolated virus, how did Dr Drosten obtain the full virus genome to select his primer sequences, and which, or so he claims, were specific to SARS-CoV-2 only? As he states in the paper:
In addition to information on reagents, oligonucleotides and positive controls, laboratories working under quality control programmes need to rely on documentation of technical qualification of the assay formulation as well as data from external clinical evaluation tests. The provision of control RNA templates has been effectively implemented by the EVAg project that provides virus-related reagents from academic research collections [18]. SARS-CoV RNA was retrievable from EVAg before the present outbreak; specific products such as RNA transcripts for the here-described assays were first retrievable from the EVAg online catalogue on 14 January 2020 (https://www.european-virus-archive.com). Technical qualification data based on cell culture materials and synthetic constructs, as well as results from exclusivity testing on 75 clinical samples, were included in the first version of the diagnostic protocol provided to the WHO on 13 January 2020.
Notice the careful wording. He doesn't actually say that he obtained SARS-CoV-2 from these sources, but later acknowledges:
We gratefully acknowledge the authors, the originating and submitting laboratories for their sequence and metadata shared through GISAID2, on which this research is based. All authors of data may be contacted directly via www.gisaid.org: National Institute for Viral Disease Control and Prevention, China CDC (Wenjie Tan, Xiang Zhao, Wenling Wang, Xuejun Ma, Yongzhong Jiang, Roujian Lu, Ji Wang, Weimin Zhou, Peihua Niu, Peipei Liu,Faxian Zhan, Weifeng Shi, Baoying Huang, Jun Liu, Li Zhao, Yao Meng, Xiaozhou He, Fei Ye, Na Zhu, Yang Li, Jing Chen, Wenbo Xu, George F. Gao, Guizhen Wu); Wuhan Institute of Virology, Chinese Academy of Sciences (Peng Zhou, Xing-Lou Yang, Ding-Yu Zhang, Lei Zhang, Yan Zhu, Hao-Rui Si, Zhengli Shi); Institute of Pathogen Biology, Chinese Academy of Medical Sciences and Peking Union Medical College (Lili Ren, Jianwei Wang, Qi Jin, Zichun Xiang, Yongjun Li, Zhiqiang Wu, Chao Wu, Yiwei Liu); and National Institute for Communicable Disease Control and Prevention (ICDC), China CDC (Zhang Y-Z, Wu, F, Chen Y-M, Pei Y-Y, Xu L, Wang W, Zhao S, Yu B, Hu Y, Tao Z-W, Song Z-G, Tian J-H, Zhang Y-L, Liu Y, Zheng J-J, Dai F-H, Wang Q-M, She J-L and Zhu T-Y)
Stil doesn't mention the source of the particular SARS-CoV-2 sequence. Let's dig deeper, here's the WHO protocol from January 13th:
We acknowledge the originators of sequences in GISAID (www.gisaid.org): National Institute for Viral Disease Control and Prevention, China, Institute of Pathogen Biology, Chinese Academy of Medical Sciences, Peking Union Medical College, China, and Wuhan Jinyintan Hospital Wuhan Institute of Virology, Chinese Academy of Sciences, China). We acknowledge Professor Yong-Zhen Zhang, Shanghai Public Health Clinical Center & School of Public Health, Fudan University, Shanghai, China for release of another sequence (MN908947).
The virus RNA of, allegedly, SARS-CoV-2 was therefore derived from a database upload in GISAID by Chinese researchers, and Dr Drosten subsequently created the first commercially available RT-PCR screening kit based on this genome. Is this the genome?
https://www.ncbi.nlm.nih.gov/nuccore/MN908947?%3Fdb=nucleotide
The primer sequences that Dr Drosten chose, and which he claims are specific to SARS-CoV-2, are not found in that genome.
Now let's look at something funny at Wikipedia, here's the section describing isolation and sequencing of SARS-CoV-2, emphasis mine:
By 12 January 2020, five genomes of SARS-CoV-2 had been isolated from Wuhan and reported by the Chinese Center for Disease Control and Prevention (CCDC) and other institutions;[15][106] the number of genomes increased to 42 by 30 January 2020.[107]
It's unusual that Wikipedia would accept "other institutions" as a source. Let's follow the footnotes:
https://platform.gisaid.org/epi3/start/CoV2020
Doesn't link to a particular genome, requires registration, web-archived link isn't a deep link but requires same registration.
https://virological.org/t/novel-2019-coronavirus-genome/319
Doesn't link to a particular genome but to ongoing uploads, and discussions about sequencing errors. More on this later.
https://nextstrain.org/narratives/ncov/sit-rep/de/2020-01-30
Isn't actually listing genomes, but a geographical map.
Which exact genome sequence of SARS-CoV-2 was Dr Drosten using on January 13th when he picked the primer sequences? The paper doesn't provide any reference for SARS-CoV-2, but strange enough, does list identifiers for SARS-CoV-1 and other controls.
A research team from the University of Graz collected random sea water from the Aral sea in Kazakhstan, and ran popular RT-PCR screening kits against the sea water. See page 5:
https://digital.csic.es/bitstream/10261/217720/1/Highly%20matching_Mora.pdf
The authors conclude:
Consequently, we argue that, although primers for detecting SARS-CoV-2 and other short sequence matches were found, their finding does not support that the virus was actually present in these samples. We rather detected naturally occurring relatives or Coronavirus-like sequences of which most are of yet unknown origin. The latter is more plausible as we implemented a DNA-based approach. Nevertheless, the fact that Coronavirus-like sequences and even short exact matches to SARS-CoV-2 were detected, hints at the presence of yet unknown environmental viruses or related sequences in this exemplarily analyzed plant-associated habitat. Considering that we also found SARS-CoV-2 test-primer and -probe matches, the existence of such sequences provides a possible explanation for the occurrence of false positive tests as recently reported by news outlets (18, 19) that were welcomed by conspiracy theorists.
Some of the primer sequences are found in the human genome itself, which this website explains away as a fundamental misunderstanding of how RT-PCR works:
Which still raises the question how specific the primer sequences are to SARS-CoV-2, and why the protocol by Institut Pasteur would claim that those sequences are from the SARS-CoV-2 genome when in fact they are from human chromosome 8. Sloppy writing, when the fate of humankind was depending on the rapid rollout of these test kits?
Let's shed some light on the reliability of genome sequencing itself. This page shows the first pack of sequences from isolated virus:
https://virological.org/t/preliminary-phylogenetic-analysis-of-11-ncov2019-genomes-2020-01-19/329
Notice the caveats, emphasis mine:
The number of genetic differences in the genomes is close to the error rate of the sequencing process. Some of the observed differences may be artefacts of this process.
and, from another scientist, emphasis mine: [see comments]
"Since I believe ~ 50% of the diversity in the tree comes from sequencing errors, the TMRCAs would likely be even more recent - possibly pushing the interval towards the end of December. That means that the outbreak was detected almost immediately after the first case, which - given that this is flu season in China - is just amazing. Detecting an outbreak of pneumonia (similar to flu) of a novel coronavirus that fast is truly impressive."
Therefore we have established that not only are at least some commercial RT-PCR kits for screening throat swabs for traces of SARS-CoV-2 RNA developed in the absence of isolated live virus, but the sequencing of - allegedly - live SARS-CoV-2 for the purpose of sharing the genome with other scientists across the globe is prone to sequencing errors. As RT-PCR is based on very short primer sequences, and as the Mora paper has found, even random sea water from Kazakhstan can and will yield false positives for primers. The discussion whether the RT-PCR method itself is scientifically sound is misguided. The question should rather be: how scientific is the process of finding specific primers that match mutations of SARS-CoV-2 and nothing else?
Finally, let's hear the Washington Post describe the initial testing chaos in the US, emphasis mine:
Shortcomings with the tests were first noticed in late January, after the CDC sent an initial batch to 26 public health labs across the country. According to those with knowledge of what unfolded, false-positive reactions emerged at 24 of the 26 labs that first tried out the kits in advance of analyzing samples gathered from patients.
“Only two of them got it right,’’ said a senior federal scientist who reviewed the development of the kits and internal test documentation, and who concluded that the false positives were caused by contamination that occurred at the CDC.
The false positives arose during testing of “negative control’’ samples that contained highly purified water and no genetic material. That aspect of testing was essential to confirm that the test results were reliable and not because of contamination.
And this is how the FDA solved the issue, emphasis mine:
The new recommendations focus on “high-complexity testing laboratories” that are certified under federal rules known as Clinical Laboratory Improvement Amendments. This group of facilities includes many hospital labs, like the one where epidemiologist Michael Mina works at Brigham and Women’s Hospital. “Essentially it’s opening up a clear and concise avenue for labs like the one at Brigham and Women’s,” Mina says. “It’s what I’ve been advocating for a month now.”
If these labs want to make and use their own tests, FDA says, they should send five positive and five negative samples, as determined by their protocols, to another qualified lab for confirmation. FDA still requires labs to submit what’s known as an emergency use authorization (EUA) application to the agency. “For a reasonable period of time after validation and while they are preparing their EUA requests, FDA does not intend to object to the use of these tests for specimen testing,” the recommendations state.
“It’s their method of saying just go ahead and start doing the testing, it’s sort of got out of hand, and if you don’t hear from us in a year, just keep testing,” Mina says.
I guess this explains a lot.





Your demonstration of evidence for cycle threshold abuse will be useful for me, as I had planned to re-cover the PCR Ct issue to help further demonstrate why the care home deaths were indeed murders - not simply sped up C19 deaths - by giving readers a crash course on the follies of PCR.
It is, as you say, a 'done' and stale subject within the lockdown critical crowd, so I had been putting off putting out such an article until I could devise some means to also make it particularly useful to readers, so an early coverage by WATN would be good.
Time to also look into Kary Mullis's death, Aug 2019.